TL;DR:
- Genetic risk assessment turns hidden DNA information into actionable health decisions.
- It involves family history review, gene testing, and professional counseling for personalized results.
- The process helps manage hereditary cancer, medication responses, and family planning options.
Most people assume cancer or serious illness is something that happens to others, not something quietly written into their DNA. Yet 5–10% of cancers are linked to inherited genetic variants, and millions of people carry those variants without ever knowing it. Genetic risk assessment changes that equation. It turns an invisible biological blueprint into clear, actionable health information. Whether you are wondering about hereditary cancer, trying to find the right medication without trial and error, or preparing to start a family, understanding your genetic risk is one of the most practical health decisions you can make.
Table of Contents
- What is genetic risk assessment?
- Evaluating hereditary cancer risk: Process and challenges
- Genetic risk assessment for medication response (pharmacogenomics)
- Genetic risk assessment in family planning: Expanded carrier screening
- What most people miss about genetic risk assessment
- Explore your genetic risk assessment options with Gene Matrix
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Defines genetic risk assessment | Genetic risk assessment uses personal and genetic data to estimate your risk for certain diseases, medication responses, or inherited conditions. |
| Hereditary cancer screening | Modern panels and counseling can identify high-risk hereditary cancer cases and offer early intervention options. |
| Medication optimization through genetics | Pharmacogenomics helps tailor medication choices to your genetic makeup, reducing adverse reactions and improving efficacy. |
| Family planning made safer | Expanded carrier screening detects common risk factors for inherited disorders before pregnancy, supporting informed family planning. |
| Importance of expert guidance | Personalized counseling is crucial for understanding results and taking meaningful actions beyond the test report. |
What is genetic risk assessment?
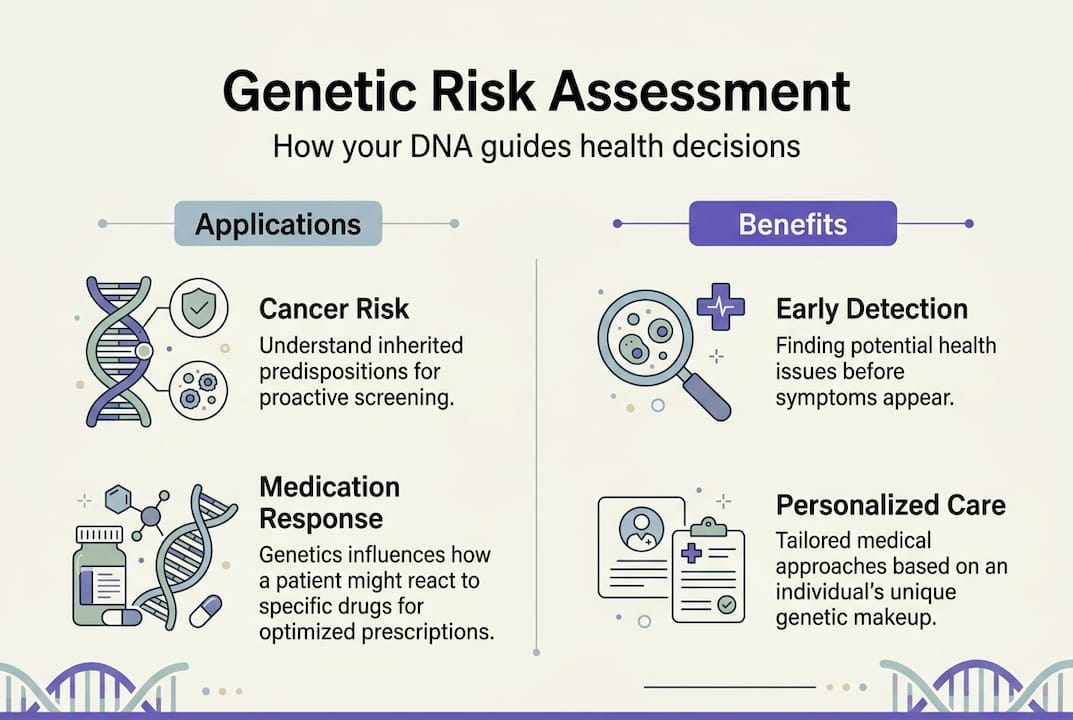
Genetic risk assessment estimates your individual risk for hereditary diseases, cancers, medication reactions, or the chance of passing an inherited condition to your children. It is not a crystal ball. It is a structured process that combines your family history, laboratory testing, and expert interpretation to give you a clearer picture of what your biology may be telling you.
The process typically moves through three core stages:
- Family history review: A trained counselor maps out at least three generations of your family's health records, looking for patterns of early-onset cancer, recurrent diseases, or unexplained deaths.
- Genetic panel testing: Next-generation sequencing (NGS) panels scan multiple genes simultaneously, making it far more efficient than older single-gene tests. This is where the global impact of genetic testing becomes visible, as faster and broader panels catch risks that would have been missed a decade ago.
- Genetic counseling: Before and after testing, a certified genetic counselor helps you understand what results mean and, more importantly, what to do with them.
"The goal of genetic risk assessment is not to predict the future with certainty, but to give individuals enough information to make informed, proactive choices about their health."
One thing that surprises many people: a negative result does not always mean zero risk. Rapid genetic testing has improved dramatically, but no panel catches every possible variant. That is why counseling is not optional. It is the part of the process that turns a lab report into a personal health strategy. Think of the test as the data and the counselor as the interpreter who makes that data usable. For a broader look at what testing involves, the genetic testing overview at Gene Matrix is a solid starting point.
Evaluating hereditary cancer risk: Process and challenges
Hereditary cancer risk is probably the most well-known application of genetic risk assessment, and for good reason. 5–10% of cancers are caused by inherited germline variants, meaning the mutation is present in every cell of your body from birth. For some syndromes, the stakes are very high. People with hereditary breast and ovarian cancer (HBOC) syndrome face a lifetime risk up to 80% for breast cancer, and Li-Fraumeni syndrome carries a risk approaching 100% for multiple cancer types.
The evaluation process starts with a three-generation pedigree, a detailed family health map that flags red-flag patterns: multiple relatives with the same cancer, early-onset diagnoses (before age 50), or rare cancers like male breast cancer. From there, multigene NGS panels test for BRCA1, BRCA2, Lynch syndrome genes (MLH1, MSH2, MSH6, PMS2), TP53, PALB2, and many others in a single run.
| Approach | Genes tested | Time | Best for |
|---|---|---|---|
| Single-gene test | 1 | Fast | Known family mutation |
| Multigene NGS panel | 10–100+ | Moderate | Unknown or complex history |
| Polygenic risk score (PRS) | Thousands of variants | Moderate | Population-level risk |
Here is where it gets nuanced. Not every result is a clean positive or negative. Variants of uncertain significance (VUS) are genetic changes that labs cannot yet classify as harmful or harmless. Incomplete penetrance means that even if you carry a high-risk variant, you may never develop the associated cancer. These edge cases are exactly why family genetic testing tips and pre-test counseling matter so much. You need to understand what you are signing up for before you see the results.
For conditions like skin cancer, genetic risk can also inform surveillance strategies, even when the primary mutation is not inherited. The genetic testing technology behind modern panels continues to evolve, making results more precise each year.
"A positive result is not a diagnosis. It is a signal to act earlier and smarter."
Pro Tip: If a close relative has already tested positive for a hereditary mutation, ask about single-site testing first. It is faster, less expensive, and tells you exactly what you need to know.
Genetic risk assessment for medication response (pharmacogenomics)
Here is a fact that most people never hear from their doctor: your genes significantly influence how you respond to medications. Pharmacogenomics (PGx) is the branch of genetic risk assessment that maps this relationship. Variants in CYP450, DPYD, and HLA genes predict how your body metabolizes drugs and whether you are at risk for serious adverse reactions.
The CYP450 enzyme family is responsible for breaking down a huge portion of common medications. If you are a "poor metabolizer" of a drug, it builds up in your system and causes toxicity. If you are an "ultra-rapid metabolizer," the drug clears too fast to work. Neither scenario is obvious without testing. And 6–10% of the population may benefit directly from genotype-guided dosing adjustments.
The drug categories most affected by PGx variants include:
- Antidepressants and antipsychotics: SSRIs, SNRIs, and tricyclics are heavily influenced by CYP2D6 and CYP2C19 variants.
- Cancer therapies: Drugs like 5-fluorouracil carry serious toxicity risk in patients with DPYD deficiency.
- Anticoagulants: Warfarin dosing is directly linked to CYP2C9 and VKORC1 variants.
- Antiepileptics: HLA-B*15:02 predicts severe skin reactions to carbamazepine in certain populations.
- Pain management: Codeine metabolism varies widely based on CYP2D6 status.
| Gene | Drug category | Clinical impact |
|---|---|---|
| CYP2D6 | Antidepressants, opioids | Dosing and efficacy |
| DPYD | Chemotherapy (5-FU) | Toxicity risk |
| HLA-B*15:02 | Antiepileptics | Severe skin reactions |
| VKORC1 | Warfarin | Bleeding risk |
The AI-powered genomics benefits of modern PGx analysis mean that results can now be cross-referenced against your full medication list, flagging interactions before they become problems.
Pro Tip: Before starting a new psychiatric medication or chemotherapy regimen, ask your provider specifically about pharmacogenomic testing. A single test can inform decisions across dozens of future prescriptions.
Genetic risk assessment in family planning: Expanded carrier screening
Carrier screening is one of the most underutilized tools in reproductive health. You can carry a single copy of a recessive genetic variant and have zero symptoms, yet still pass that variant to your child. If both parents are carriers of the same condition, each pregnancy carries a 25% chance of the child being affected.
Carrier frequency is at least 1 in 100 for most screened conditions, which means this is not a rare concern. ACOG and ACMG both recommend pan-ethnic carrier screening for all prospective parents, regardless of ancestry, because NGS panels now test for over 100 genes covering recessive and X-linked disorders.
Conditions commonly included in expanded carrier screening:
- Cystic fibrosis (CFTR gene)
- Spinal muscular atrophy or SMA (SMN1 gene)
- Fragile X syndrome (FMR1 gene)
- Sickle cell disease (HBB gene)
- Tay-Sachs disease (HEXA gene)
Here is a practical step-by-step approach for prospective parents:
- Schedule pre-conception genetic counseling. Ideally, this happens before pregnancy, giving you time to understand your options.
- Both partners get tested. Carrier status only matters when both parents carry the same variant.
- Review results with a counselor. If both partners are carriers, you will discuss options including preimplantation genetic testing (PGT) during IVF, prenatal diagnosis, or natural conception with monitoring.
- Document results for future pregnancies. Carrier status does not change, but your family planning decisions might.
"Knowing your carrier status before pregnancy gives you choices. Finding out during pregnancy narrows them."
If you want a clear walkthrough of what to expect before your appointment, the DNA testing preparation guide covers the process in practical detail.
What most people miss about genetic risk assessment
After working with genetic data across thousands of cases, one pattern becomes clear: people focus almost entirely on the test result and almost not at all on what comes after. A positive BRCA2 finding or a high polygenic risk score is not the end of the story. It is the beginning of a very personal decision-making process.
The technology behind genetic testing research has advanced to the point where results arrive in days. But emotional readiness, understanding probabilities rather than certainties, and building an individualized action plan, those take longer. A result without a plan is just anxiety with a lab report attached.
What actually changes outcomes is what you do with the information. Increased surveillance, prophylactic interventions, medication adjustments, or informed family planning decisions. Generic risk scores mean very little without a counselor helping you translate them into your specific life context. The real value of genetic risk assessment is not the data. It is the personalized roadmap that follows.
Explore your genetic risk assessment options with Gene Matrix
Understanding your genetic risk is one thing. Acting on it with the right support is another.

At Gene Matrix, our CLIA-certified platform combines AI analysis trained on over 500,000 genetic profiles with expert counseling support, so your results come with context, not just data. Whether you are exploring hereditary cancer risk, optimizing your medication response through our GenePGx module, or preparing for parenthood with expanded carrier screening, our genetic testing research and science-backed approach guide every step. Check out our DNA testing subscriptions to find the right panel for your needs, or download the Genetic health app to manage your genomic health in one place.
Frequently asked questions
What diseases can genetic risk assessment help predict?
Genetic risk assessment can help estimate risk for hereditary cancers, medication responses, and inherited disorders. NGS panels now test for over 100 inherited conditions, including cystic fibrosis and spinal muscular atrophy.
How accurate are genetic risk assessments?
Accuracy varies by panel and condition. Typical panels detect 50–80% of hereditary risks, but variants of uncertain significance and false negatives mean results always require professional interpretation.
What should I expect during a genetic risk assessment?
You will complete a family history review, undergo genetic panel testing, and receive counseling before and after your results. The full process is designed to ensure you understand both the findings and your options.
Can genetic risk assessment influence medication choices?
Yes. Pharmacogenomic testing identifies variants that predict drug metabolism and adverse reactions, allowing providers to adjust doses or choose safer alternatives before problems arise.
Is expanded carrier screening recommended for everyone?
Major organizations including ACOG and ACMG recommend pan-ethnic carrier screening for all prospective parents, regardless of family background or ancestry.
