TL;DR:
- Genetic screening provides a structured, science-backed way to understand health risks and inherited conditions.
- Preparation, including family history and risk assessment, is crucial for accurate and meaningful testing outcomes.
- Clinical testing with counseling ensures accurate interpretation and supports informed health decisions.
Uncertainty about inherited health risks or unexpected medication reactions leaves many families stuck, unsure whether to act or wait. Genetic screening removes that guesswork. It gives you a structured, science-backed path to understanding your cancer risk, your body's response to medications, and the health landscape your children may inherit. But the process only delivers real value when you follow it correctly, from gathering your family history to interpreting a result with a trained counselor. This guide walks you through every stage so you can make informed, confident decisions about your health and your family's future.
Table of Contents
- Preparing for genetic screening: Requirements and risk assessment
- Step-by-step: The genetic screening process
- Understanding methodologies and accuracy in genetic screening
- Interpreting results and next steps: From variants to action
- Why clinical pathways and counseling matter in genetic screening
- Get expert-guided genetic screening with Gene Matrix
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Risk assessment is key | Start with a thorough review of your personal and family health history for accurate genetic screening. |
| Follow clinical protocols | Seek pre- and post-test genetic counseling for best interpretation and risk management. |
| Technology affects accuracy | Clinical gene panels and laboratory methods offer greater precision than DTC screening. |
| Interpret results carefully | Variants and uncertain findings should be reviewed by professionals and may require family cascade testing. |
| Ethical considerations matter | Protect your privacy and psychological well-being by choosing reputable clinical providers. |
Preparing for genetic screening: Requirements and risk assessment
Before any sample is collected or any gene is analyzed, preparation is everything. The quality of your results depends heavily on the information you bring into the process. Skipping this stage is one of the most common mistakes people make, and it often leads to incomplete testing or mismatched panels.
The first thing you need is a thorough personal medical history. That means documenting any prior diagnoses, surgeries, biopsies, or cancer treatments. Equally important is your family history, specifically a three-generation account that includes grandparents, parents, siblings, aunts, uncles, and cousins. Risk assessment via detailed personal and 3-generation family history forms the foundation of genetic screening and directly shapes which tests are ordered.
Here is what to compile before your first appointment:
- Full names and relationships of blood relatives
- Diagnoses and ages at diagnosis for any cancer, heart disease, or neurological condition
- Ethnic background, since some variants are more prevalent in specific populations
- Reproductive history, including miscarriages or birth defects
- Prior genetic test results, if any family member has been tested
This information feeds directly into your genetic risk assessment, which determines your pre-test probability and guides test selection. A high-risk family history, for example, may warrant a broad multi-gene panel rather than a single-gene test.
| Screening trigger | Recommended action |
|---|---|
| First-degree relative with BRCA1/2 | Order hereditary breast/ovarian cancer panel |
| Two or more relatives with colorectal cancer | Evaluate for Lynch syndrome testing |
| Multiple medications with poor tolerance | Consider pharmacogenomic (PGx) panel |
| Child with developmental concerns | Pediatric genomic evaluation |
For families with complex histories, family genetic testing tips can help you organize information before your counseling appointment. The NCCN guidelines also provide clear criteria for who qualifies for hereditary cancer testing.
Pro Tip: Call relatives before your appointment and ask specific questions about diagnoses and ages. A vague "someone had cancer" is far less useful than "my maternal aunt was diagnosed with ovarian cancer at 48."
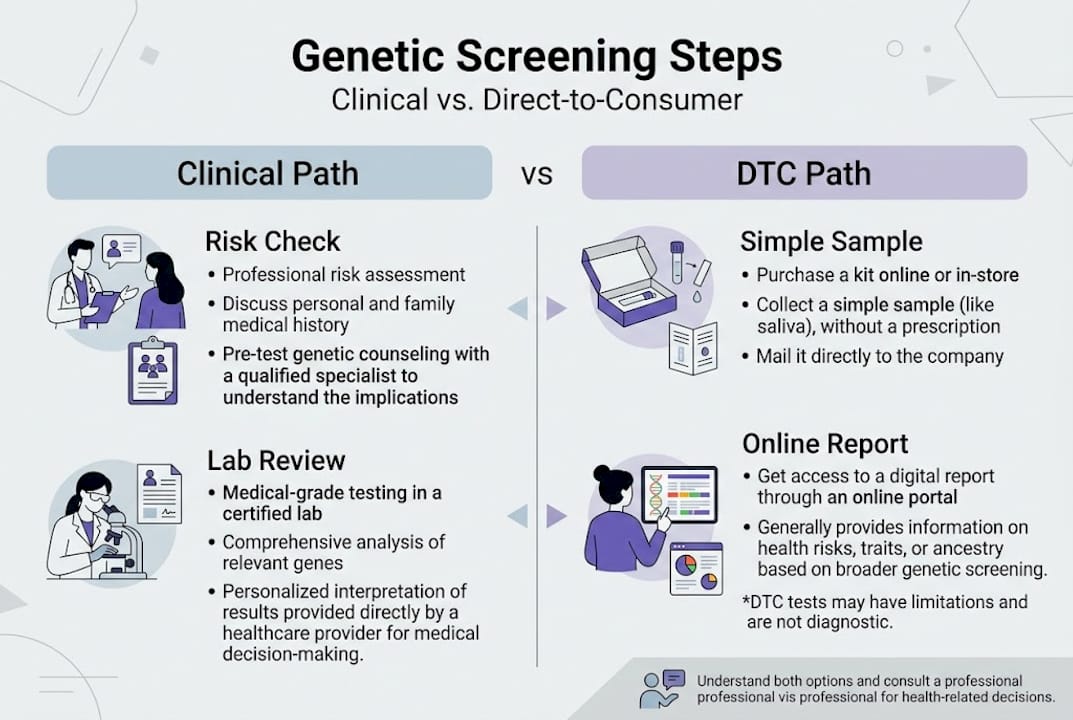
Step-by-step: The genetic screening process
Once your preparation is complete, the actual screening process follows a clear sequence. Each step builds on the last, and skipping any one of them increases the risk of misinterpretation.
- Pre-test genetic counseling. A certified genetic counselor reviews your risk profile, explains what the test can and cannot detect, and helps you choose the right panel. This step is not optional in a clinical setting.
- Informed consent. You review and sign documentation that covers what happens to your sample, how results are shared, and your rights regarding data privacy.
- Sample collection. Most clinical tests use a saliva swab or blood draw. Saliva is common for hereditary panels; blood may be preferred for certain pharmacogenomic or somatic tests.
- Laboratory analysis. Your sample goes through next-generation sequencing (NGS), polymerase chain reaction (PCR), or targeted genotyping depending on the panel. NGS panels cover 40+ genes across more than ten cancer types.
- Result generation. The lab produces a structured report classifying variants as pathogenic, likely pathogenic, variant of uncertain significance (VUS), or negative.
- Post-test counseling. A counselor walks you through your results, explains what they mean clinically, and outlines next steps. The recommended process always includes this final counseling stage for interpreting results accurately.
For a broader look at how this fits into personalized care, the precision medicine guide offers helpful context. You can also explore the genetic testing science behind clinical-grade panels.
| Feature | Clinical testing | Direct-to-consumer (DTC) |
|---|---|---|
| Genes analyzed | 40+ via NGS | Limited SNPs |
| Counseling included | Yes | Rarely |
| Insurance coverage | Often eligible | Typically not |
| Regulatory oversight | CLIA-certified labs | Variable |
| Actionability | High | Low to moderate |
Pro Tip: Ask your provider specifically whether the lab is CLIA-certified. That certification means the lab meets federal standards for accuracy and quality, which matters enormously when results could influence treatment decisions. Understanding gene panel testing can also help you ask smarter questions before you start.
Understanding methodologies and accuracy in genetic screening
Knowing the steps is one thing. Understanding the technology behind them helps you evaluate what your results actually mean and how much confidence to place in them.
Clinical genetic screening relies primarily on next-generation sequencing (NGS), which reads millions of DNA fragments simultaneously. This allows labs to analyze dozens of genes in a single test run. Clinical NGS panels cover more than 40 genes, while DTC tests are limited to selected single-nucleotide polymorphisms (SNPs), and DTC false positives run around 40%.

Pharmacogenomic (PGx) testing is a specialized branch that examines genes like CYP2D6, DPYD, and TPMT to predict how your body metabolizes specific medications. Pharmacogenomic testing is highly accurate and can prevent dangerous drug reactions before they happen. This is especially relevant for psychiatric medications, chemotherapy agents, and blood thinners.
Here is a breakdown of key methodologies and what they detect:
- Multi-gene hereditary cancer panels: Detect pathogenic variants in BRCA1, BRCA2, MLH1, MSH2, and dozens of other cancer-risk genes
- Pharmacogenomics panels: Map drug metabolism pathways to optimize prescribing
- Polygenic risk scores (PRS): Aggregate hundreds of common variants to estimate population-level risk
- Multi-cancer early detection (MCED): Blood-based tests that screen for cancer signals across multiple organ types
Accuracy varies significantly by method. The positive predictive value (PPV) for MCED tests sits at 43% in high-risk populations, meaning not every positive signal is a true cancer. PRS detects roughly 40% of clinically significant prostate cancer cases. These numbers are not reasons to avoid testing. They are reasons to use rapid genetic testing within a clinical framework where a counselor can contextualize the results.
The personalized medicine benefits of combining these methodologies are well documented, and AI-powered genomics platforms are now making multi-method analysis faster and more accessible.
| Methodology | Genes/markers covered | Typical accuracy |
|---|---|---|
| Clinical NGS panel | 40+ genes | >99% analytical sensitivity |
| DTC SNP genotyping | Selected SNPs only | ~60% (40% false positive rate) |
| Pharmacogenomics | CYP2D6, DPYD, TPMT, others | High (clinically validated) |
| MCED blood test | Multi-cancer signal | 43% PPV in high-risk groups |
Interpreting results and next steps: From variants to action
Receiving your results is not the finish line. It is the starting point for action. Understanding what each result classification means will help you avoid two common traps: panic and false reassurance.
Results typically fall into one of four categories:
- Pathogenic or likely pathogenic: A variant known or strongly suspected to increase disease risk. This warrants immediate follow-up with a specialist.
- Variant of uncertain significance (VUS): A change detected in a gene, but without enough evidence to classify it as harmful or benign. VUS results are common and require cascade family testing and ongoing monitoring.
- Negative (no variant detected): The tested genes showed no known pathogenic variants. This reduces but does not eliminate risk.
- Benign or likely benign: A variant present but not associated with increased disease risk.
"A negative result does not mean zero risk. It means no known pathogenic variant was found in the genes tested. Residual risk remains based on family history and lifestyle factors."
For pathogenic results, your next steps typically include specialist referral, enhanced surveillance (such as annual MRI for BRCA carriers), preventive medications, or surgical risk reduction. Cascade testing, meaning offering testing to first and second-degree relatives, is one of the most powerful tools in genetic testing for prevention because it can identify at-risk family members before symptoms appear.
Ethical considerations matter here too. Privacy, discrimination, and psychological impact are real concerns, particularly around insurance and employment. In the U.S., GINA (Genetic Information Nondiscrimination Act) provides some protections, but gaps exist. Always confirm results, whether from a DTC or clinical source, through post-test counseling before making any major medical decisions.
Pro Tip: Before sharing results with family members, speak with your genetic counselor about the best way to communicate findings. Some relatives may not want to know, and that choice deserves respect.
Why clinical pathways and counseling matter in genetic screening
Most guides focus on the mechanics of genetic screening. What they miss is the human layer underneath it, and that layer is where real outcomes are won or lost.
Clinical testing is preferred over DTC for both accuracy and counseling, while DTC testing empowers users but carries real risk of misinterpretation and privacy exposure. That distinction is not just technical. A VUS result without counseling can cause months of unnecessary anxiety. A negative result without context can create false confidence that leads someone to skip surveillance they actually need.
Cascade screening is another area where clinical pathways deliver outsized value. When one family member tests positive, systematic testing of relatives can prevent cancers before they develop. That prevention chain only happens when a clinical team is coordinating the process. DTC platforms do not offer that infrastructure.
The psychological dimension of genetic screening is also underestimated. Learning you carry a BRCA2 variant or a Lynch syndrome mutation is not just a data point. It reshapes how you think about your body, your children, and your future. Clinical settings provide access to mental health support and genetic counselors trained to navigate those conversations. Clinical accuracy matters, but so does the support system around the result. Choosing clinical vs DTC testing is ultimately a choice between a guided process and an isolated one.
Get expert-guided genetic screening with Gene Matrix
You now understand the full arc of genetic screening, from preparation through interpretation. The next step is putting that knowledge into action with a team that can guide you through every stage.

Gene Matrix is a CLIA-certified, AI-powered genomics platform built for exactly this purpose. With panels covering hereditary cancer, pharmacogenomics, pediatric health, and nutrigenomics, Gene Matrix delivers actionable reports within 72 hours, backed by AI trained on 500,000+ genetic profiles. Their genetic testing technology ensures clinical-grade accuracy, and their genetic innovation team stays ahead of evolving science. Whether you are screening for BRCA variants, optimizing medications, or planning for your family's future, Gene Matrix provides the clinical structure and expert support that turns results into real health decisions.
Frequently asked questions
What information do I need before starting genetic screening?
You need your personal medical details and a three-generation family health history, including cancer diagnoses, ages at diagnosis, and other major conditions in blood relatives.
Is direct-to-consumer genetic screening reliable?
DTC false positives run around 40%, and DTC tests lack the counseling and clinical validation found in certified laboratory testing, making them less reliable for medical decision-making.
What happens if my genetic test returns a variant of uncertain significance (VUS)?
VUS results are common and typically require cascade family testing and ongoing review with a genetic counselor to clarify the variant's clinical significance over time.
How does genetic screening help with medication choices?
Pharmacogenomic screening tests genes like CYP2D6 and DPYD that control drug metabolism, enabling physicians to prescribe medications at the right dose with fewer adverse reactions.
Are there privacy risks with genetic screening?
Genetic screening does carry privacy and discrimination risks, so selecting a CLIA-certified clinical provider with clear data protection policies is essential before you begin.
